 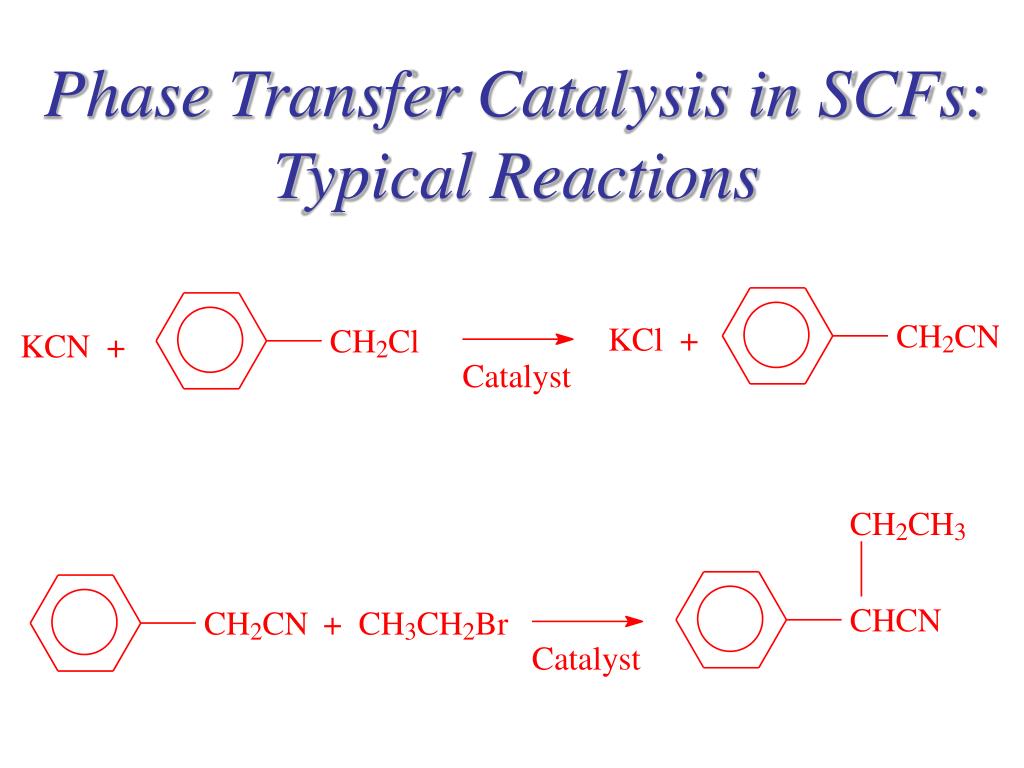
The solvent front was measured to be 31 mm in length.TLC Sample Product Rf (mm) Reactant 1 Rf (mm) Reactant 2 Rf (mm)0 minutes 0 2/31 = 0.06 10/31 = 0.3210 minutes 18/31 = 0.58 4/31 = 0.13 8/31 = 0.2630 minutes 21/31 = 0.68 6/31 = 0.19 11/31 = 0.3560 minutes 25/31 = 0.81 4/31 = 0.13 9/31 = 0.29 After the sample was recovered from the reaction vial, the methylene layer was removed from the upper aqueous phase and transferred to a sample vial containing anhydrous calcium chloride. The results from the TLC tests conducted on spots from the reaction solution taken at 0 minutes, 10 minutes, 30 minutes, and a final 60 minutes are listed below. With the use of the catalyst, benzyl-n-butylammonium chloride, to move naphtholate anions from the aqueous to the organic phase so that reaction involving allyl bromide would proceed. Madison McVe圜HEM 237 – 549November 17, 2016Phase Transfer Catalyst Lab ReportResults and Discussion:In this experiment, students identified the phase transfer catalyst and its ability to move a reactant from the aqueous to the organic state in order for the reaction to take place. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
